
Considering 70 kg weight for a person, not affected by sex or age, this means 1.1 g of sulfur per day. The recommended dietary allowance (RDA) for sulfur has been estimated to be 13-14 mg/kg of body weight per day. The amount of sulfur compounds in food greatly varies depending on the type of food: 8% for egg white, 5% for beef as well as for chicken and fish, and 4% for dairy products and plant proteins. The sulfur compounds contained in food are amino acids or vitamins including methionine (Met), cysteine (Cys), homocysteine (HCy), cystine (Cys-Cys), taurine (Tau), lipoic acid, thiamine, and biotin as well as the glucosinolates and allylic sulfur compounds that are contained in cabbage and cauliflower (cruciferous vegetables). It is doubtful whether other amino acid derivatives or other “-onium” compounds could play this role: quaternary ammonium compounds are unable to effectively methylate acceptor compounds, and oxonium compounds, such as a hypothetical oxygen analogue of SAM, would produce such a powerful methylating agent that it would methylate cellular nucleophiles without the need for an enzyme. In cell metabolism, a sulfonium compound such as S-adenosylmethionine (SAM) mediates most biochemical methylation reactions. These compounds undergo sequential oxidation to sulfoxides (R 2SO) and sulfones (R 2SO 2), conferring to these derivatives novel unexpected roles. 
Thioethers can form sulfonium ions (R 3S +) by donating electrons to other organic species thanks to their ability to sink electrons and stabilize a negative charge on a neighboring carbon. The thioether (R 2S) moiety of Met is more reactive than the analogue ether (R 2O). 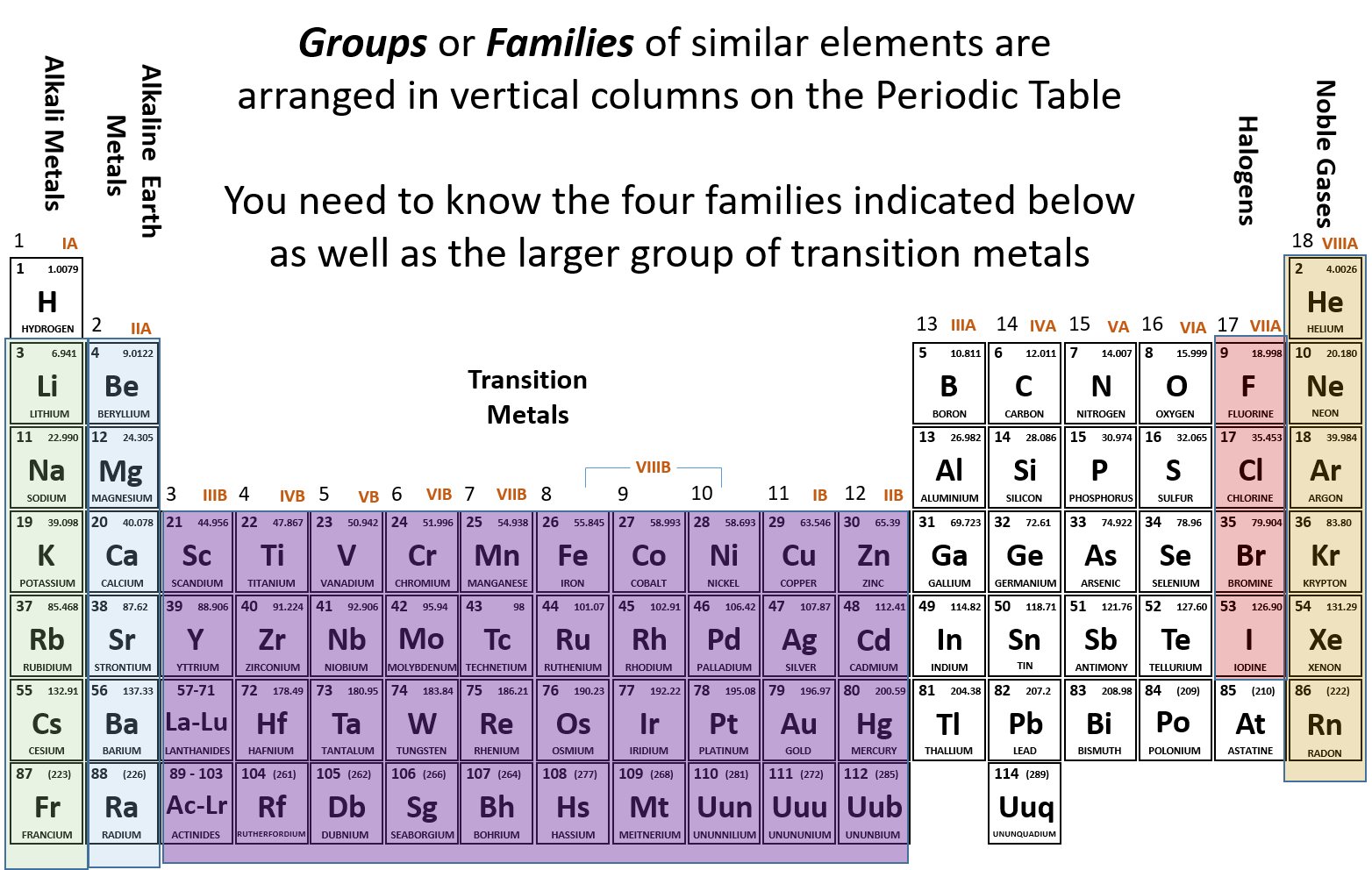
The increased atomic size confers to sulfur a lower electronegativity than oxygen. Sulfur has unique characteristics that differentiate it from oxygen. Actually, sulfur and oxygen belong to the same group in the periodic table however, Met and Cys analogues with the sulfur atom replaced by oxygen do not serve the same function. Noteworthy, the bulk of biomolecules consists only of carbon, hydrogen, nitrogen, and oxygen atoms, and the presence of sulfur accounts for the distinctive properties of sulfur compounds. Sulfur belongs to chalcogens, elements of the 16 group of the periodic table, which display the awesome characteristic of having a variety of redox states and redox potentials allowing them to form interchalcogen bonds and atom exchange reactions, giving rise to a vast number of sulfur species that take part in biological processes. In living organisms, sulfur is one of the most fundamental elements and the seventh most abundant mineral in the human body. We will describe the chemistry and the biochemistry of well-known metabolites and also of the unknown and poorly studied sulfur natural products which are still in search for a biological role. This review will explore sulfur metabolism related to redox biochemistry and will describe the various classes of sulfur-containing compounds spread all over the natural kingdoms. The chemical properties, the large number of oxidation states, and the versatile reactivity of the oxygen family chalcogens make sulfur ideal for redox biological reactions and electron transfer processes. Sulfur metabolism in humans is very complicated and plays a central role in redox biochemistry. However, some higher species such as humans are able to build new sulfur-containing chemical entities starting especially from plants' organosulfur precursors. Animals are not able to fix inorganic sulfur into biomolecules and are completely dependent on preformed organic sulfurous compounds to satisfy their sulfur needs. Terrestrial organisms such as plants and microorganisms are also able to incorporate sulfur in organic molecules to produce primary metabolites (e.g., methionine, cysteine) and more complex unique chemical structures with diverse biological roles. It is no coincidence that marine organisms are one of the most important sources of sulfur natural products since most of the inorganic sulfur is metabolized in ocean environments where this element is abundant. 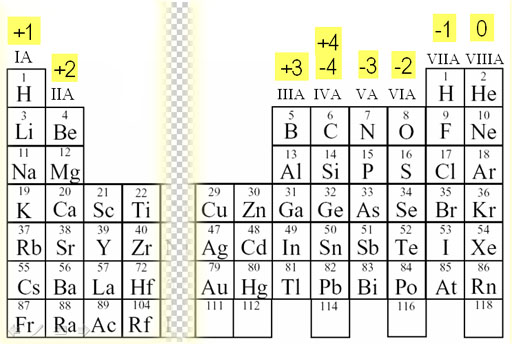
Sulfur natural compounds are utilized by all living beings and depending on the function are distributed in the different kingdoms. Sulfur contributes significantly to nature chemical diversity and thanks to its particular features allows fundamental biological reactions that no other element allows.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
